  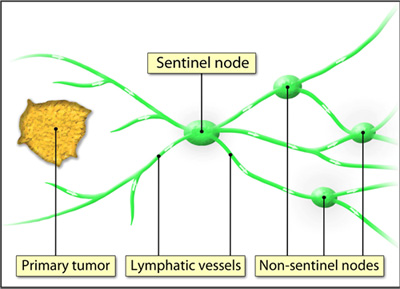
We hypothesized that SLNB would identify patients with nodal metastases with acceptable sensitivity. We therefore prospectively evaluated the performance characteristics of SLNB using indocyanine green (ICG) specifically in patients with clinical stage I disease with intermediate- and high-grade histologic subtypes. 13Īdditional trials of SLNB followed by lymphadenectomy as the reference standard are needed to inform practice in EC. Of published studies that have evaluated SLNB predominantly in this patient population, most were retrospective and performed at a single center, 10, 11, 12 did not perform PALND, 10, 11, 12 or used technetium Tc 99m or blue dye rather than more contemporary tracers. Only 13% of patients in the Sentinel Node and Endometrial Cancer (SENTI-ENDO) trial, 7 28% in the Determining the Sensitivity of Sentinel Lymph Nodes Identified With Robotic Fluorescence Imaging (FIRES) trial, 8 and 49% in the Pelvic Sentinel Lymph Node Detection in High-Risk Endometrial Cancer (SHREC) trial 9 had high-grade histologic subtypes. 4, 5, 6Īlthough SLNB has gained acceptance in the context of low-grade EC, its role in high-grade EC remains unclear. 
4, 5 Theoretically, SLNB should reflect the status of the entire nodal basin and provide the pathologic information required to guide decisions on adjuvant therapy while avoiding the heightened risks of intraoperative injury, chronic lymphedema, and other complications associated with complete lymphadenectomy. Sentinel lymph node biopsy (SLNB), or resection of only the first nodes receiving lymphatic drainage from the tumor site, has therefore been proposed as a less invasive strategy for nodal assessment. 1 Nodal metastases are traditionally identified on pelvic lymphadenectomy (PLND) and para-aortic lymphadenectomy (PALND), but 2 randomized clinical trials 2, 3 have suggested that lymph node resection independent of the effect of adjuvant therapy does not improve survival in patients with EC. Role of the Funder/Sponsor: The funding sources had no role in the design and conduct of the study collection, management, analysis, and interpretation of the data preparation, review, or approval of the manuscript and decision to submit the manuscript for publication.Įndometrial cancer (EC) that has metastasized to surrounding lymph nodes is associated with a poor prognosis and requires administration of adjuvant therapy. Dr Cusimano is supported by the Canadian Institutes of Health Research Vanier Canada Graduate Scholarship and American College of Surgeons Resident Research Scholarship. No other disclosures were reported.įunding/Support: This study was supported by the Princess Margaret Cancer Foundation of Princess Margaret Cancer Centre (Toronto, Ontario) and the McArthur Research Fund of the Department of Obstetrics and Gynecology at the University of Toronto (Toronto, Ontario). Supervision: Vicus, Pulman, May, Covens, Rouzbahman, Ferguson.Ĭonflict of Interest Disclosures: Dr Clarke reported receiving personal fees from AstraZeneca outside the submitted work. Statistical analysis: Cusimano, Maganti, Kupets, Zia, Ene.Īdministrative, technical, or material support: Cusimano, Bernardini, May, Hogen, Covens, Kupets, Clarke, Cesari, Turashvili, Zia, Ene, Ferguson. Ferguson, MD, Division of Gynecologic Oncology, University Health Network/Sinai Health Systems, 700 University Ave, Room 6-911, Toronto, ON M5G 1Z5, Canada ( Contributions: Drs Ferguson and Cusimano had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.Ĭoncept and design: Cusimano, Covens, Rouzbahman, Ferguson.Īcquisition, analysis, or interpretation of data: All authors.ĭrafting of the manuscript: Cusimano, Covens, Kupets, Rouzbahman, Ferguson.Ĭritical revision of the manuscript for important intellectual content: All authors. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
